
Homologue domains were created from one or more sequence blocks as defined in the Methods. Blue boxes: homologue domains of vertebrate (lamprey, chicken, and frog) LV precursors. Red: (right) brackets map the location of each structural domain, defined by X-ray crystallography, within the sequence of lamprey LV (four β:LV β sheets single α:LV α helical domain). Color code: Green: (top) LV, lamprey LV precursor sequence (reference sequence) (right) brackets denote the location within the precursor of each processed polypeptide chai n of lamprey LV: LV-1n, LV-1c, and LV-2. The amino acid sequence position for the motifs within the three LV precursors is indicated along the right hand edge of the black box. Amphipathic β strands are marked by black bars (A) and positively charged amphipathic α helixes by gray bars (B). Amphipathic motifs identified by the program composite LOCATE in the amino acid sequences of the vertebrate LV precursors (fLV, frog cLV, chicken and LV, lamprey) are plotted within the black box. Mapping of homologue domains, amphipathic motifs and known lamprey LV structural elements to the amino acid sequences of vertebrate (lamprey, chicken, frog) LV precursors. microsomal triglyceride transfer protein (MTP)įig.N-terminal domain of apolipoprotein B has structural homology to lipovitellin and microsomal triglyceride transfer protein: a “lipid pocket” model for self-assembly of apoB-containing lipoprotein particles. 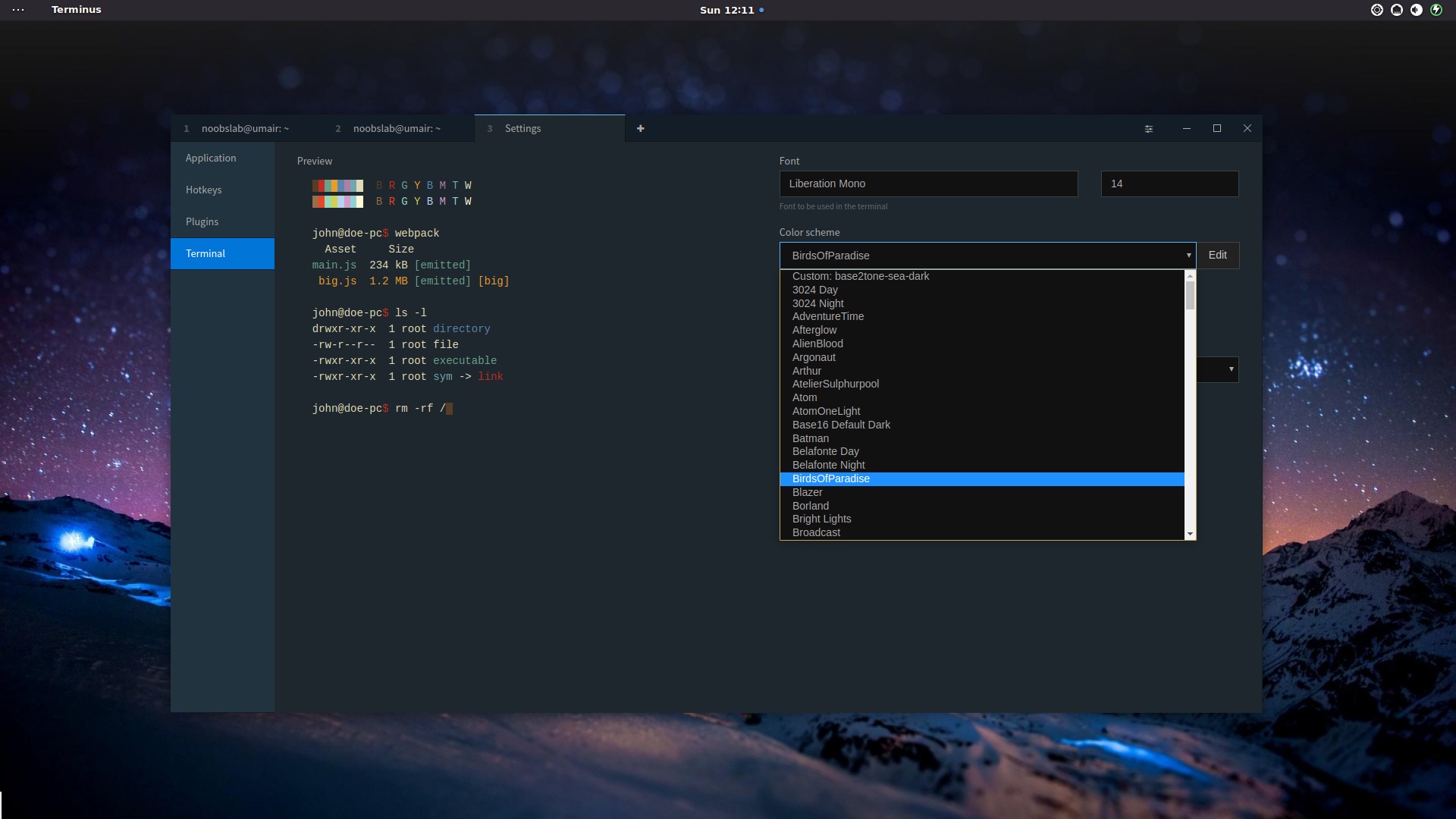
This intermediate produces a lipid nidus required for assembly of apoB-containing lipoprotein particles pocket expansion through the addition of amphipathic β strands from the β 1 domain of apoB results in the formation of a progressively larger high density lipoprotein (HDL)-like, then very low density lipoprotein (VLDL)-like, spheroidal lipoprotein particle.-Segrest, J. We also show that most of the α 1 domain of human apoB-100 has sequence and amphipathic motif homologies to human microsomal triglyceride transfer protein (MTP), a protein required for assembly of apoB-containing lipoproteins.īased upon these results, we suggest that an LV-like “proteolipid” intermediate containing a “lipid pocket” is formed by the N-terminal portion of apoB alone or, more likely, as a complex with MTP. Here we report that the first 1000 residues of human apoB-100 (the α 1 domain plus the first 200 residues of the β 1 domain) have sequence and amphipathic motif homologies to the lipid-binding pocket of lamprey lipovitellin. The X-ray crystal structure of lamprey lipovitellin is known to contain a “lipid pocket” lined by antiparallel amphipathic β sheets. elegans appeared on the list of candidate proteins. During a recent database search for protein sequences that contained similar amphipathic β strands to apoB-100, four vitellogenins, the precursor form of lipovitellin, an egg yolk lipoprotein, from chicken, frog, lamprey, and C. The process of assembly of apolipoprotein (apo) B-containing lipoprotein particles occurs co-translationally after disulfide-dependent folding of the N-terminal domain of apoB but the mechanism is not understood.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
